They have the advantage of showing the structure of a chemical just from the name, but a disadvantage of usually being cumbersome and difficult to write or speak. Scientific, or more properly systematic names, are newer names given by the IUPAC. The reason multiple names are employed for the same chemical is because each has its own advantages and disadvantages which matter differently to different groups of people. See this article for a list of the full names of the elements.Most chemicals will have both a modern scientific and older common name which are used. Square brackets around an entire number indicate the mass number of the most stable isotope. The uncertainties in the last given decimal are in parentheses. The following table lists the standard atomic weights. it becomes clear that the confusion is created by too many international committees addressing the same, basically very simple, problem of definition. IUPAC also defines standard atomic weight, but adds recommended to its definition, that is, IUPAC defines standard atomic weight as recommended relative atomic mass, which suggests that the recommended value may change in the future when more accurate data become available.įrom reading Ref. Hence, according to IUPAC's definition, the relative atomic mass is the dimensionless version of the standard atomic weight defined above. The ratio of the average mass of the atom to the unified atomic mass unitĪlthough it is not stated explicitly in the Goldbook what is meant by "average mass", it is likely and plausible that the averaging is over different isotopes weighted by terrestrial isotopic abundance. Relative atomic mass (atomic weight), A r On the other hand, the official IUPAC publication, IUPAC Goldbook, defines: Thus A r(X) is the numerical value of m(X) when m(X) is expressed The relative atomic mass A r(X) of an elementary particle, atom, or more generally an entity X, is defined by A r(X) = m(X) / m u, where m(X) is the mass of X. 
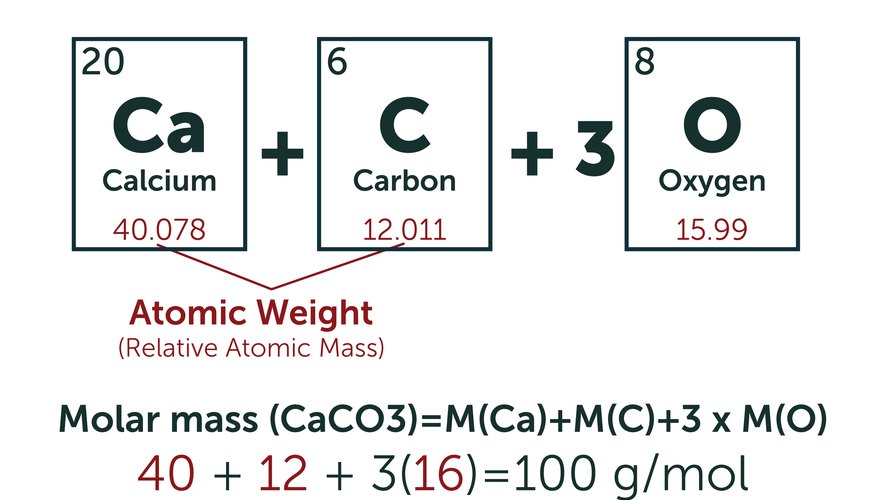
This usage is followed by Mohr and Taylor, who state that (the atomic mass constant m u is a twelfth of the mass of 12C): Relative Atomic Mass (of the isotope): A r(X), where X is an isotope We followed NIST, see the NIST web site, where clearly and unambiguously the relative mass is defined of an isotope. The concept of "relative atomic mass" is in principle a simple one, yet there is some confusion about its definition. For instance, the HCl molecule has standard atomic weight 1.00794 + 35.453 = 36.461, which is the value used in almost all chemical calculations. By using averaged masses the chemist accounts for the fact that different isotopes occur in nature. (For historical reasons the term "weight" is still used here.) In most of practical chemistry the standard weight is used as "the" mass of an element. The atomic mass averaged over isotopic abundances is called the standard atomic weight. Of all the chlorine atoms occurring on earth 75.78 % is of the lighter kind, while 24.22 % is the heavier isotope. It has two stable isotopes: 35Cl (with a mass of 34.96885271 u) and 37Cl (with a mass of 36.96590260 u). In off-the-shelf chemicals the concentrations of different isotopologues are determined by the terrestrial natural abundances of the isotopes. In most of practical chemistry different isotopologues-always present in "off-the-shelf" chemicals-are of no concern whatsoever. 
Hydrogen chloride, for instance, would be seen as a mixture of the following isotopologues: H– 35Cl, D– 35Cl, H– 37Cl, and D– 37Cl. In these fields it is common to consider the samples as mixtures of of different isotopologues, in much the same way as when the sample consists of different compounds. That is, one can distinguish the spectral peaks arising from the different isotopologues, (same molecule, different isotopic composition) in the sample. In high resolution spectroscopy and mass spectrometry masses of isotopes are observed in the spectra. 
In practice there are two ways of dealing with the different masses of isotopes: the isotope 13C is the dimensionless number 13.0033548378. It has two stable isotopes and one radioactive-but long-lived-isotope. For example, look at the element carbon (atomic number Z = 6, i.e., 6 protons). So, different isotopes of a given element have the same charge but differ in mass. Recall first that different isotopes of an element have different numbers of neutrons and the same number (the atomic number Z) of protons. 3.1 Standard Atomic Masses of the Elements.3 Standard Atomic Weights of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
